Review of the Exceptional Circumstances Framework
Pharmac is reviewing its Exceptional Circumstances Framework to make it simpler and clearer to better meet the needs of people who rely on it to get funded access to medicines.

The full text of the discussion document follows. You can read it here or download a PDF version to review offline.
Download a PDF version of the discussion document [PDF, 2.3 MB]
Our feedback form is hosted on a consultation platform.
Complete the feedback form(external link)
Frequently Asked Questions about the Exceptional Circumstances Framework and Current Review
If you have questions or want to submit feedback by email contact us on:
Table of contents
Exceptional Circumstances Framework – pathways
Named Patient Pharmaceutical Assessment (NPPA)
Executive Summary
Pharmac is taking this opportunity to update and improve the Exceptional Circumstances Framework (the Framework), which includes the Named Patient Pharmaceutical Assessment (NPPA) policy and other ways Pharmac can fund medicines for individuals in special or exceptional clinical situations.
Exceptional Circumstances Framework
Under the Pae Ora (Healthy Futures) Act 2022, Pharmac is required to have a system, or framework, that allows people funded access to medicines not listed on the Pharmaceutical Schedule (or listed on the Schedule for a different indication) when their situation is exceptional.
During the review of the Framework, there will be no changes to how Pharmac assesses applications under the current Framework. Any changes to the Framework will only take place after the review is complete and will be introduced in planned stages to ensure a smooth transition.
Everyone with an existing named patient approval will stay under their current funding arrangements regardless of the outcome of the review.
What is the Framework?
The Exceptional Circumstances Framework explains how people may be able to access funded medicines when the standard Pharmaceutical Schedule funding process does not meet their needs. It is intended to support people with unusual or complex clinical circumstances by setting out the pathways that may be available to them.
The Framework recognises that different funding pathways serve different purposes. The Schedule process supports access for population groups, while other pathways exist to respond to exceptional situations. Together, these processes are designed to work alongside each other so that people can achieve the best possible health outcomes.
The Framework includes the NPPA Policy for people with exceptional clinical circumstances, Special Authority waivers for people who broadly meet the intent of funded criteria but cannot meet every requirement, and other processes that allow Pharmac to consider unique or exceptional situations. These pathways aim to provide flexibility and clarity for people and their clinicians when standard funding rules do not apply.
How is it funded?
Funding for medicines approved under the Framework comes from Pharmac’s overall budget for community and hospital medicines. There isn’t a separate ring-fenced fund for the pathways within the Framework, instead, it draws from the same fixed medicine budget that Pharmac uses to fund medicines listed on the Pharmaceutical Schedule.
What’s included in the review?
The review will look at how the current Framework, including all the funding pathways, is working in practice and where improvements are needed. It will look at what is working well and what could be changed, removed or added, to make things better.
As well as feedback received during consultation, we will also consider the feedback people have already given us to make sure the review reflects what matters most to people.
Why is Pharmac doing the review?
Pharmac is reviewing the Exceptional Circumstances framework to ensure it is simpler, clearer, and better meets the needs of people who rely on it to get funded access to medicines. The review is needed to give clinicians a process that is straightforward and easy to navigate, and to give Pharmac clearer criteria and documentation for making timely, consistent decisions while fully meeting legislative requirements.
What is Pharmac seeking to understand?
This discussion document is part of a broader review of the Framework. Pharmac wants your feedback to help improve the Framework. We’re especially interested in hearing:
- how the Framework works for you in practice
- if or how it fits with Pharmac’s statutory objectives and priorities
- if it ensures fair access to medicines for people whose clinical needs cannot be met through the Pharmaceutical Schedule
- any gaps, barriers, or unintended consequences you have observed
- suggestions for improving clarity, transparency and consistency in decision making.
How Pharmac will engage
The review will happen in two stages. In the first stage, Pharmac will gather information on what could be improved. In the second stage, Pharmac will consult again on any proposed changes or improvements informed by the feedback received.
To make sure we are thorough and inclusive, Pharmac will use a range of methods to gather feedback and insights, (see Table One). Consideration will also be given to accessibility needs, for example, different languages, plain English and alternate formats including audio, Easy Read, large print, and New Zealand Sign Language.
|
How you can provide feedback |
Description |
|---|---|
|
Webinars * |
Join live online sessions to learn more and ask questions. |
|
Online feedback forms |
Fill out the online survey questions to share your views. The discussion document will be available to view including links to other information, videos and reading materials. |
|
|
An invitation will be sent out with an opportunity to send us your feedback by email. |
|
Workshops |
Take part in hands-on sessions for shared problem-solving. |
|
Visits to identified stakeholders |
Pharmac will attend meetings offsite, with times and venues to suit the stakeholder groups. |
|
Direct online conversations and focus groups |
One-on-one or small group discussions for detailed insights. |
|
Virtual town hall |
Join an open online meeting where you can share your views and hear from others. |
|
Social media |
Respond to quick polls or questions on Pharmac’s social media channels. |
* What is the difference between a webinar and a virtual town hall? A webinar is a live online session where information is shared about the review. It usually includes a presentation and time for questions at the end. A virtual town hall is a more open online meeting focused on discussion. You can share your views, ask questions, and hear what others think in real time.
How decisions will be made
Pharmac wants to be open about how decisions will be made as part of this review. All feedback Pharmac receives during the consultation will be carefully read and considered. We will look for common themes, different views, and any issues that people raise about what is working well and what could be improved.
After the first stage of consultation a summary of feedback will be shared with Pharmac’s Senior Leadership Team (SLT). We may also seek advice from specialist groups or people with relevant expertise where this would help us better understand specific issues and feedback received. Following the second round of consultation, there will be further decisions made at SLT. The Pharmac Board will approve the final decisions about any changes and improvements to the policy. Once the review is complete, any changes will be implemented following a clear plan to ensure a smooth transition and keep everyone informed.
Introduction
Overview of Pharmac’s role
Pharmac was set up in 1993 to provide nationally consistent access to medicines while limiting the rapid growth in expenditure occurring at the time. Pharmac's objective is to secure, for eligible people in need of pharmaceuticals, the best health outcomes that are reasonably achievable from pharmaceutical treatment. It must do this within the fixed budget set by government (the medicines budget).
To achieve its objective, Pharmac manages the Pharmaceutical Schedule (the Schedule) of publicly funded medicines that applies consistently within New Zealand. In addition, Pharmac is responsible for managing national contracts for some medical devices used in public hospitals, ensuring equitable access and cost effectiveness across the health system.
The Factors for Consideration (FFC) are Pharmac’s primary decision-making framework and apply to all decisions, including exceptions. The FFC helps Pharmac assess each funding application against Pharmac's statutory objective
Overview of the Exceptional Circumstances Framework
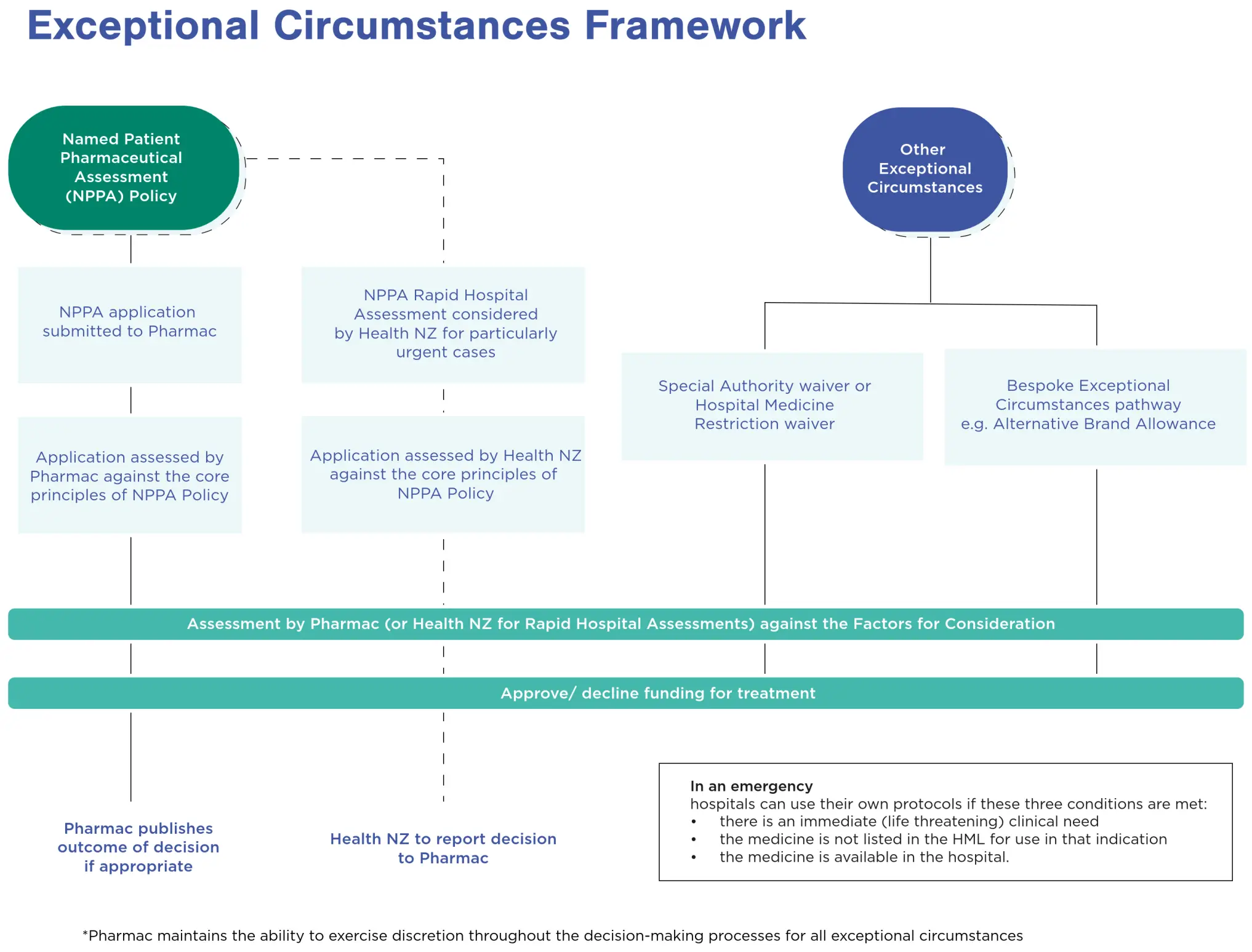
The Exceptional Circumstances Framework (see Figure One) explains the options available when someone needs medicine funding that does not fit the usual Schedule process. It is designed to help ensure people with exceptional health needs can still access medicines in certain situations. The Framework brings together several pathways, including the NPPA Policy, different types of waivers, requests for alternative brands, and processes that may allow funding for some unapproved medicines when appropriate. These pathways aim to provide clear, flexible options so people can get timely access to the medicines they need.
The need to revise the Framework and NPPA policy was identified in the government response to the review of Pharmac in 2022, which directed Pharmac to ‘give high priority to considering equitable outcomes in its decision-making processes and to further develop its analytical frameworks and tools …to support equitable outcomes’.
Scope of the review
Pharmac is reviewing the Framework as a whole. Every component of the Framework will be considered, including its principles, criteria, processes, and implementation in practice. The review is not limited to specific issues and is intended to ensure the Framework is effective, equitable, and aligned with Pharmac’s strategic direction and legislative responsibilities.
Medical devices are not within the scope of this current review. However, we welcome comments or feedback, which may be used to inform consideration of a potential future exceptions pathway or process for medical devices.
Overall, the aim of the Framework review is to:
- review the current NPPA policy and other pathways within the Framework
- determine if the existing Framework adequately meets its legislative function
- improve people's knowledge and understanding of the Framework
- strengthen the Framework and its application
- improve how rare disorders are considered and addressed within the Framework
- increase transparency of the NPPA process to reduce the percentage of applications received that do not meet the principles
- establish policies to reflect processes that have been established due to need (the use of the Framework to manage applications for alternative brands is a good example of this).
What we’ve heard so far
Pharmac has already received informal feedback from a wide range of stakeholders including consumers, clinicians, NPPA advisory panel members, Pharmac staff, and others impacted by the Framework. This feedback will be carefully considered, alongside the feedback that will be gathered through this consultation. We will also directly engage with clinicians who don’t currently use the process, including those working in primary and rural care and clinicians in the regions where applications are fewer.
Together these insights will help ensure the Framework is effective, equitable, and aligned with Pharmac’s strategic direction and legislative responsibilities.
Key themes from feedback received to date include:
- the Framework is not fit for purpose and often misunderstood
- uncertainty about how applications are assessed and when NPPA funding is impacted by Schedule applications
- lack of clarity around what constitutes “exceptional” and how the policy is interpreted to determine this
- confusion about decision-making processes and a lack of transparency
- equity barriers, particularly affecting people with high health needs and other priority populations
- lack of clear guidance and feedback on the application process and decisions made
- inconsistent processes for approving alternative brand allowances.
More detail on the feedback we've received so far
Rare disorders
Rare Disorders are defined as those affecting one person in every 2,000 or fewer. Ultra-rare disorders are even less common, occurring in one person in every 50,000 or fewer. Funding for medicines for people with rare disorders is considered through the Schedule or through NPPA applications. The main difference is that the Exceptional Circumstances Framework applies to individual people, while rare disorders relate to small groups of people with the same condition. This helps make sure there are clear ways for both individuals and small patient groups to access the treatments they need.
However, even when a condition is classified as rare, it can still affect a relatively large number of people. In some cases, this means the condition may not meet the criteria for NPPA. When this happens, Pharmac considers the group, rather than as individual cases. Although the condition is rare, the total number of patients may be large enough for Pharmac to gather evidence and expert advice through the standard Pharmaceutical Schedule process. At present, there is no specific pathway under the Exceptional Circumstances Framework for funding medicines for rare disorders.
Rare Disorders Strategy | Ministry of Health website(external link)
Download questions as a PDF [400KB](external link)
Exceptional Circumstances Framework – pathways
There are several pathways within the Exceptional Circumstances Framework. These pathways allow people funded access to medicines not listed on the Pharmaceutical Schedule (or listed on the Schedule for a different indication) when their situation is exceptional.
The Exceptional Circumstances Framework
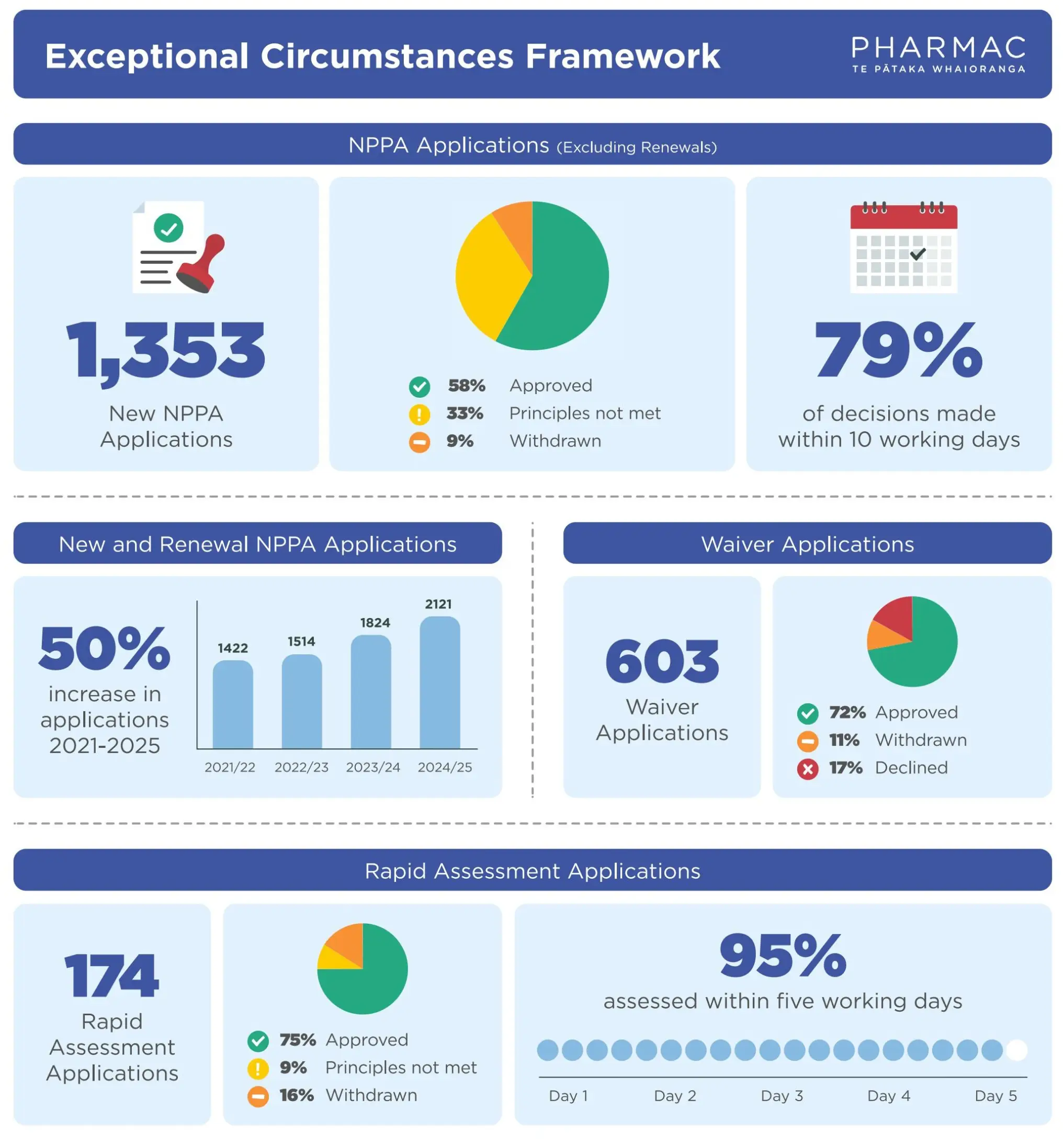
The infographic (Figure 2) summarises key information about the use of the Exceptional Circumstances pathways, offering a clear snapshot of the volume and nature of applications.
Named Patient Pharmaceutical Assessment (NPPA)
For people with exceptional clinical circumstances
Pharmac developed the NPPA policy to guide how it makes decisions about funding medicines for people in exceptional circumstances. It is designed for people who have urgent and unusual clinical needs that are not met by treatments currently funded through the Schedule. Before applying for NPPA, all suitable funded treatments must have been tried. If there are other funded options available, they need to have been used and/or be clinically unsuitable for the person’s specific situation.
How NPPA works
Pharmac (or Health New Zealand hospitals, in urgent hospital cases) uses three core NPPA Principles to decide whether a treatment should be considered for funding through the NPPA process. If an application meets these Principles, it will then be assessed using Pharmac’s FFC.
Pharmac can also decide that a different funding process might be more suitable for a particular application. Even if an application does not meet the NPPA policy principles, Pharmac still has the option to fund a treatment using wider discretion if the situation is appropriate.
How Pharmac processes a NPPA application
Depending on the complexity of the application, the assessment process can involve input from several Pharmac staff and external clinical experts.
Exceptions advisors assess NPPA applications and may seek input from Pharmac Therapeutic Group Managers (TGMs). Clinical advice on individual applications may also be sought from the NPPA Advisory Panel, members of the Pharmacology and Therapeutics Advisory Committee (PTAC), a Specialist Advisory Committee, or other independent experts.
How to make a NPPA application
When NPPA is not the right pathway
This policy is not intended for treatments that have already been considered for funding through the Schedule for the same condition. It also does not apply where there is already a suitable funded treatment available or when the treatment would be suitable for many people. However, if a person’s clinical circumstances differ from those previously assessed, for example their health need is significantly different or they cannot use the currently funded treatment, this may progress for assessment under the FFC.
NPPA sets a standard
When an application is approved under this policy, it sets a precedent. This means that future applications for the same treatment, for people in similar circumstances, are also likely to be approved. This includes when a Schedule application has been received after approval under NPPA.
NPPA Rapid Assessment applications
Rapid assessments are applications for treatments used within Health New Zealand hospitals in situations that are considered clinically urgent. Pharmac commits to making decisions on such applications in less than five working days (noting that if a hospital requires a decision faster they are authorised to make the decision locally).
Financial implications for NPPA
Funding for NPPA-approved applications comes from the medicines budget with no separate or ring-fenced allocation. This means that treatments funded by both NPPA and the Schedule come from the same budget. NPPA-related expenditure has grown over time, driven by an increase in application volumes and a rise in high-cost medicines being assessed through the Framework.
Other Exceptional Circumstances
Special Authority waiver and Hospital Medicine Restriction waivers
If a person’s circumstances align with the intent of a Special Authority or Hospital Medicine Restriction waiver, but do not fully meet the stated requirements, prescribers may request that those requirements be waived.
Sometimes the usual requirements cannot be met safely or appropriately. For instance, some medicines require another treatment to be tried first. Methotrexate is one example. as it cannot be used during pregnancy, a waiver may be appropriate for people who are pregnant or planning pregnancy. These decisions typically have no fiscal impact, as funding has already been approved; the focus is on whether the person’s situation meets the intent of the group eligible for funding.
How to make a Special Authority waiver application
Bespoke exceptional circumstances pathway
Sometimes there is a need for Pharmac to change the funded brand of medicine. When this happens Pharmac understands the change may not be suitable for everyone. Currently, one way to fund alternative brands, is through a bespoke framework pathway. This allows people to remain on their current treatment when it’s not clinically suitable for them to transition to the newly funded brand.
The pathways for some applications, such as alternative brand requests for diabetes technology, are bespoke and have their own application forms and pharmaceutical-specific assessment criteria.
Other exceptional circumstances forms
Automatic approvals
The Framework also allows some medicines that are not approved by Medsafe and are not listed on the Schedule to be funded, without needing a full assessment. This is called an automatic approval. This pathway is used when a medicine is well‑established, at low risk, and commonly needed, so requiring a full assessment would add unnecessary delays. An example is the routine approval of oral vitamin A and E.
Review Approach
Pharmac is reviewing the NPPA policy and the broader Framework following longstanding calls for change. We are now able to undertake this review and are seeking feedback from stakeholders.
The aim of this consultation is to provide clear, accessible opportunities for everyone to share their views. While feedback can be given through our website and email, we know these channels may not reach all key stakeholders. To ensure broad engagement, we will also hold targeted sessions, hui, and work through trusted networks, offering flexible ways for people to participate.
The consultation period will be open for a set timeframe, although timelines (in Table Two) may be subject to change depending on the nature and volume of feedback received.
The first step is the release of this Discussion Document and an online survey. Other methods will also be used to gather feedback, for example, webinars and workshops. This stage focuses on sharing information and gathering feedback from all stakeholders. Pharmac will use this input to identify improvements and develop options. A second consultation will follow on proposed changes.
This two-stage public consultation has the expected benefits of:
- providing Pharmac with additional information that enhances analysis and decision-making process
- allows for a more open, consultative process where stakeholders can be more involved throughout the process on a sensitive subject matter
- stakeholders can feed in earlier in the policy process, reducing risks that Pharmac has pre-determined an outcome.
This approach will allow for more meaningful feedback, which should enable Pharmac to develop a solution that is robust, equitable and sustainable. Pharmac is open to seeking feedback on any aspect of the Framework from all stakeholders.
|
Stage |
Key Steps |
Estimated Time |
|---|---|---|
|
Public consultation on discussion document |
|
Consultation is open for 12 weeks. |
|
Submissions analysis |
|
8 weeks |
|
Drafting proposed changes paper |
|
4 weeks |
|
Public consultation on proposed changes paper |
|
8 weeks |
|
Submissions analysis and draft documents final decision |
|
10 weeks (note: Pharmac closed for 2 weeks over Christmas/New Year) |
|
Final decision |
|
10 weeks |
Feedback received to date
Following feedback received to date, general considerations were developed for the review include the following topics.
1. Consider whether the current Framework and the principles are fit for purpose
Currently, about a third of NPPA applications (excluding renewals) are found not to meet the three Principles used to determine whether to progress an application before they are viewed against the FFC, and a decision made by the Board or its delegate is required.
As noted, the exceptional circumstances team consulted with clinicians to understand the limitations of the application process and one of the key areas that was highlighted for improvement was to improve clinicians’ understanding of the NPPA policy. When looking at the Principles in isolation, clinicians can feel unclear about what is required or how Pharmac interprets the policy to determine what is exceptional.
The pathways for some applications, such as alternative brand requests, are bespoke and have their own application forms and pharmaceutical-specific assessment criteria. Mechanisms to manage requests for alternative brands are set up on an ad-hoc basis where it is identified that a brand change may significantly impact a patient group. The creation of a bespoke pathway is often resource intensive, with no predetermined mechanism for these pathways. It can also be difficult to assess applications under a different set of criteria each time one is created.
Applications for cancer treatment are becoming more specialised and complex and can be challenging to apply the NPPA policy too, as the range of genetic tests and targeted treatments increases. Treatments like immune checkpoint inhibitors that work well for many different types of cancer, add to the complexity.
Applications for rare disorders can also be challenging under the current NPPA policy because these conditions often affect small patient groups, resulting in limited clinical evidence to support funding decisions. Schedule applications often remain on the Options for Investment (OFI) for some time due to the high cost and limited evidence for treatment. This creates a gap where NPPA funding may be restricted, yet no Schedule application is actively considered, leaving patients with few viable pathways for access.
2. Consider societal impact
The Government’s 2025/26 Letter of Expectations suggested Pharmac include the wider fiscal impacts to Government, and broader societal and non-health outcomes, of funding medicines and medical devices. This includes assessment and decision-making to account for fiscal benefits beyond the health sector and to consider the broader economic and societal benefits of medical technologies.
Rather than heath technology assessments considering only health system perspectives and impacts, a societal approach includes all costs and benefits associated with a policy intervention. This perspective considers not only direct medical costs and health outcomes but also indirect costs and benefits such as productivity losses or gains, social care costs, and broader economic impacts on various sectors of society.
As part of this review, any changes to the Framework must align with the Government’s request for Pharmac to update its decision-making and evaluation models to consider the broader fiscal and societal impacts of funding decisions.
3. Consider inequities in the current Framework and ensure accessibility in applications to those with high health needs
Currently the NPPA policy requires applications to be submitted on a patients’ behalf by any clinician, within their scope of practice. Currently approximately 75% of applications are submitted by a secondary or tertiary care specialist (both in public and private settings). This can create inequities depending on a patient’s ability to access these services. The review will provide an opportunity to explicitly incorporate principles and/or assessment processes to ensure that those with the greatest health need are considered.
The NPPA application and assessment process doesn’t ask for patient ethnicity information; however, we are able to find this information from NHI data which can be gathered for reporting purposes. In 2022/23, a report was requested regarding the number of applications for those who identify as Māori. It was noted that a greater proportion of NPPA applications for people who identify as Māori were approved (75%) compared to the average (63%). However, it appears that there may be slightly fewer NPPA applications submitted for Māori compared to the general population. In the last financial year, 16% of NPPA and waiver applications were submitted for Māori while 17% of the population identify as Māori.
Currently Pharmac does not have data on any other priority populations but intend to include this work as part of the revision of the Framework.
Māori population estimates at 30 June 2024 | Stats NZ(external link)
4. Consider the use of discretion as part of assessing applications
The review will examine where and how discretion is currently applied in the assessment of applications, and the guidelines under which this occurs. While discretion is used in specific and limited circumstances, this is not always clearly articulated in existing guidance. As part of the review, we aim to develop clearer and more explicit guidance for consumers, applicants and Pharmac staff to support greater transparency and consistency in decision‑making. This will include clarifying the circumstances in which discretion may be applied, how it is assessed, and how this is communicated to applicants, with a view to improving understanding of the process and increasing confidence in the application of the Framework.


