Losartan potassium 50 mg with hydrochlorothiazide 12.5 mg tablets: brand change
The Miro brand of losartan with hydrochlorothiazide will be funded 1 February 2026. Arrow-Losartan & Hydrochlorothiazide will no longer be funded from 1 July 2026.
Same active ingredient, in the same quantities
The supplier of losartan potassium with hydrochlorothiazide is changing from Teva to Miro. Miro's "I-Losartan & Hydrochlorothiazide - Ipca" contains the same active ingredient, in the same quantities as Teva's brand. It should work in your body the same way.
Medsafe has reviewed "I-Losartan & Hydrochlorothiazide - Ipca". Medsafe has found that the new brand meets international standards of safety, quality, and effectiveness.
What's changing
The new brand of losartan potassium with hydrochlorothiazide was listed on 1 February 2026. The key difference is pack size – the Teva brand is a 30-pack and the new Miro brand is a 90-pack. The tablets are the same as the previous brand but in different packaging (see images below).
| Supplier | Brand name | Presentation | Pharmacode | Subsidy | Pack size |
|---|---|---|---|---|---|
| Teva | Arrow-Losartan & Hydrochlorothiazide | yellow tablet, oval, 13 mm | 2390620 | $4.31 | 30 tablets |
| Miro | I-Losartan & Hydrochlorothiazide - Ipca | yellow tablet, oval, 13 mm | 2716828 | $4.00 per 30, $7.25 per 90 | 90 tablets |
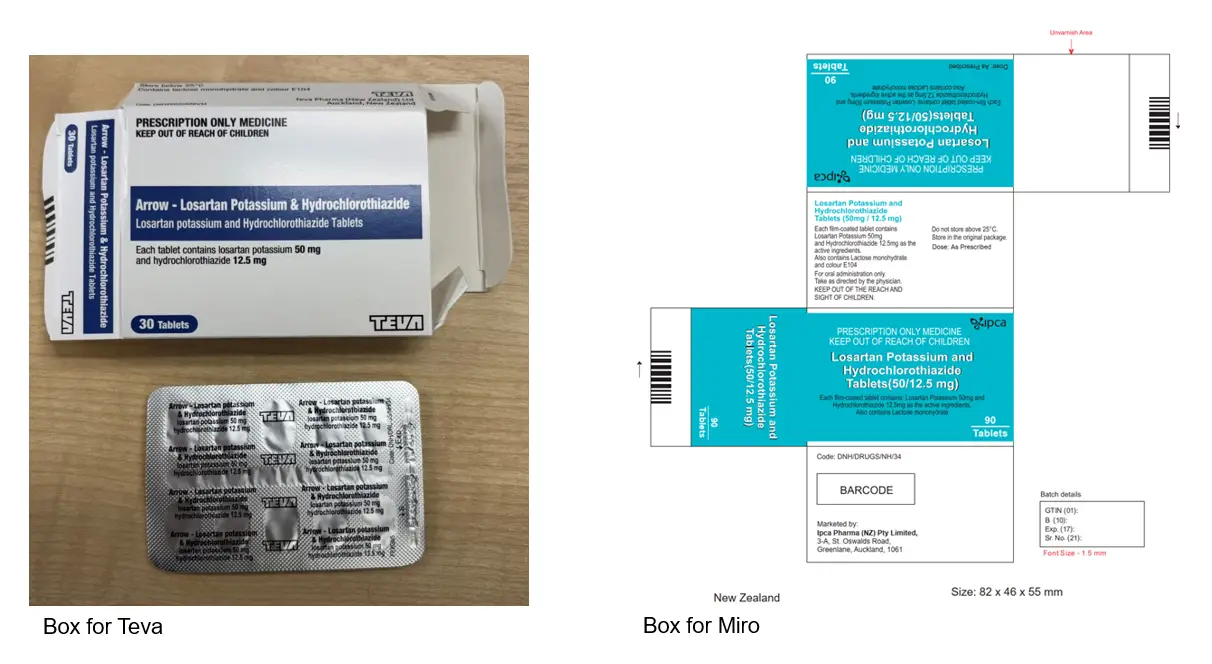

The tablets for both brands look the same, yellow and oval.
Why is this happening?
This is a tender brand change. Every year, Pharmac runs a tender process to create competition among suppliers to supply many medicines used by New Zealanders.
The tender helps Pharmac manage the costs of medicines. Any savings we make are returned to the medicines budget to help pay for new medicines.
Your brand change is helping fund more medicines for more New Zealanders.
Information for pharmacists
Miro's I-Losartan & Hydrochlorothiazide - Ipca was listed from 1 February 2026. From 1 July 2026, it will have principal supply status and Teva's Arrow-Losartan & Hydrochlorothiazide will be delisted.
Pharmacodes
- Teva's Arrow-Losartan & Hydrochlorothiazide: 2390620
- Miro's I-Losartan & Hydrochlorothiazide - Ipca: 2716828
Thank you for the time you spend supporting people through a change of brand.
Key dates
1 February 2026 | I-Losartan & Hydrochlorothiazide - Ipca listed
1 July 2026 | Arrow-Losartan & Hydrochlorothiazide delisted.
Who to contact
If you have questions about this change, email enquiry@pharmac.govt.nz
Please include as much information as you can about the product (presentation, brand, pharmacode) and who your wholesaler is.
Sign up to our email list for regular emails about supply issues and more(external link)
Pharmac is a government agency. We do not sell medicines or related products.


