Levonorgestrel: The funded brand is changing
The brand of emergency contraceptive pill (levonorgestrel) is changing from Levonorgestrel BNM to Levonorgestrel-1 (Supplied by Lupin).
What's changing
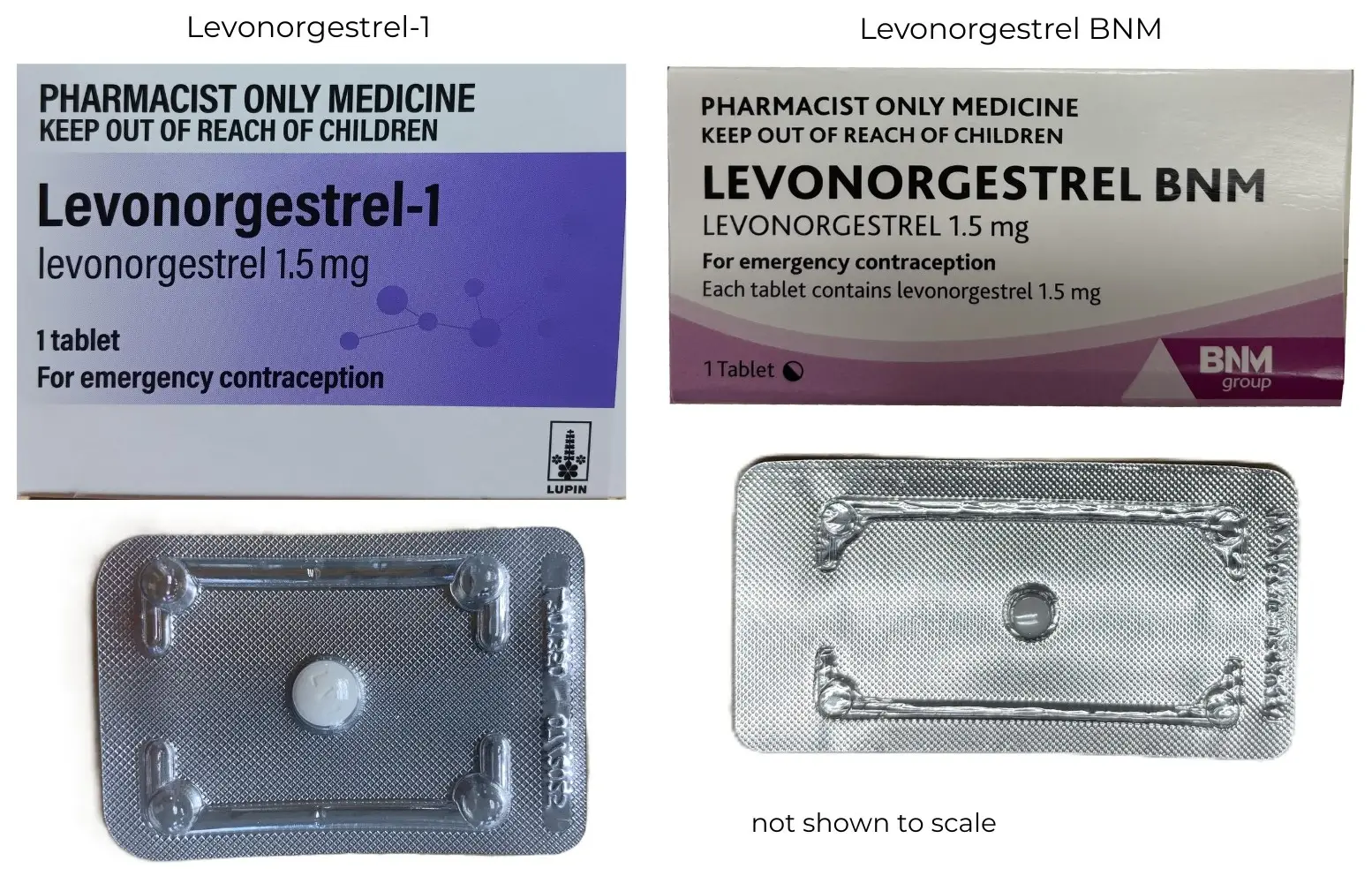
The funded brand of levonorgestrel is changing from Levonorgestrel BNM to Levonorgestrel-1, supplied by Lupin. Levonorgestrel-1 is Medsafe registered. Medsafe confirms that it is as safe and effective as the BNM brand.
The Lupin brand was reviewed by the Tender Clinical Advisory Committee. The committee found that the packaging is appropriate. The committee noted that the new tablet is slightly larger (8 mm) than the BNM tablet, which is 6 mm wide. As this is a one-off medication, we do not anticipate this to be a problem.
People who take levonorgestrel
If you have used levonorgestrel in the past as an emergency contraceptive pill, you might notice that this brand looks a little different. Medsafe has confirmed that Levonorgestrel-1 is as safe and effective as Levonorgestrel BNM.
Why is this happening?
This is a tender brand change. Every year, Pharmac runs a tender process to create competition among suppliers to supply many medicines used by New Zealanders.
The tender helps Pharmac manage the costs of medicines. Any savings we make are returned to the medicines budget to help pay for new medicines.
Your brand change is helping fund more medicines for more New Zealanders.
Pharmacists
|
Brand |
Supplier |
Presentation |
Pharmacode |
Subsidy |
Measure / Qty |
Date |
|---|---|---|---|---|---|---|
|
Levonorgestrel BNM |
BNM |
Tab 1.5 mg |
2645424 |
$1.75 |
per 1 |
Delisting date: 1 June 2026 |
|
Levonorgestrel-1 |
Lupin |
Tab 1.5 mg |
2712164 |
$1.31 |
per 1 |
Listing date: 1 January 2026 |
- Levonorgestrel (ECP) brand change | Nov 25 [PDF 1.4 MB]
Key dates
1 January 2026 | Levonorgestrel-1 funded
1 June 2026 | BNM Levonorgestrel delisted
Lupin then has principal supply status until 30 June 2028.
Your feedback on this brand change
We sought feedback about this brand change as part of our May 2025 consultation on tender brand changes. We received feedback from health professionals asking us to fund ulipristal acetate. While that was outside the scope of our consultation, ulipristal acetate is part of ongoing considerations. Funding of ulipristal acetate was also raised at the Reproductive Health and Sexual Health Committee meeting in March 2025.
March 2025 record of the Reproductive Health and Sexual Health Committee meeting [PDF, 375 KB]
Thank you to everyone who gave us feedback on the proposed brand changes.
Who to contact
If you have questions about this brand change, email enquiry@pharmac.govt.nz
Please include as much information as you can about the product (presentation, brand, pharmacode) and who your wholesaler is.
Sign up to our email list for regular emails about supply issues and more(external link)
Pharmac is a government agency. We do not sell medicines or related products.


