Iloprost (Vebulis) nebulisers: Supply issue
Supplies of Vebulis iloprost nebulisers (Pharmacode: 2643006) were low throughout New Zealand.
15 April 2026 | Stock released to wholesalers
The supplier advises that the stock has now been released to wholesalers. It can take another 1 to 2 weeks to reach pharmacies around the country.
Affected product
A significant increase in demand in December 2025 and January 2026 led to a supply issue affecting iloprost nebuliser solution.
Chemical: Iloprost
- Presentation: Nebuliser soln 10 mcg per ml, 2 ml
- Brand: Vebulis
- Pharmacode: 2643006
- Subsidy: $166.53
- Measure / Qty: per 30
Schedule listing for iloprost(external link)
Alternative product listed from 9 February
From 9 February 2026, we listed an alternative. This product is not Medsafe approved and needs to be prescribed and dispensed in line with section 29A of the Medicines Act.
- Presentation: Nebuliser soln 10 mcg per ml, 2 ml
- Brand: Vebulis S29
- Pharmacode: 2722186
- Subsidy: $166.53
- Measure / Qty: per 30
Prescribing and supplying an unapproved medicine
Section 29A of the Medicines Act 1981 allows for medicines that are not Medsafe approved to be prescribed and supplied to people.
We know supplying an unapproved medicine. In this case, however, this will allow people to continue to access an appropriate treatment.
We apologise for any inconvenience this causes.
What patients need to know about unapproved medicines – Healthify website(external link)
Same medicine, different label
This product is made by the same company and the formulation is identical. However, it is labelled for sale in Turkey. The supplier is labelling the box in English however the nebules will still be labelled in Turkish. The nebules have the same brand name "Vebulis" but all other writing is in Turkish.
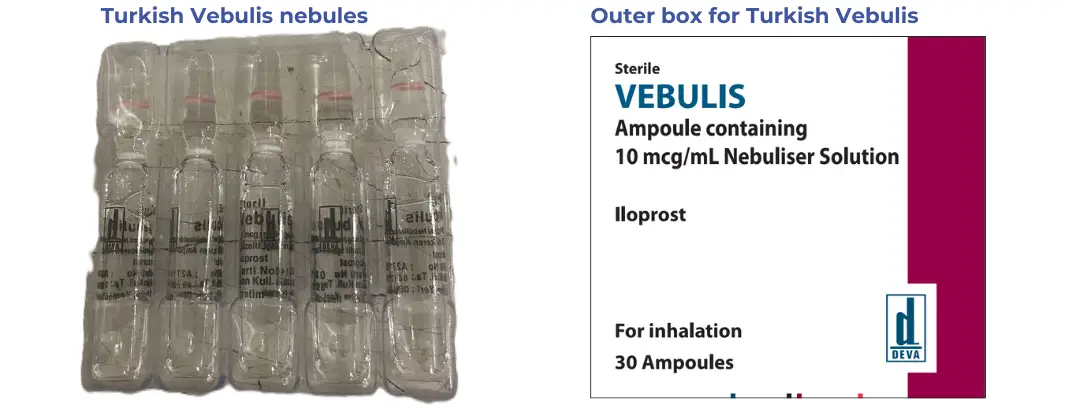
Flier for patients
The flier explains that this is the same medicine as usual but that the nebules are labelled in Turkish. It also says that their informed consent is needed for this product because it is not Medsafe approved.
- Iloprost (Vebulis) nebuliser solution supply issue [PDF 368 KB]
Resupplied April 2026
The supplier advises that the stock has now been released to wholesalers. It can take another 1 to 2 weeks to reach pharmacies around the country.
Who to contact
If you have questions about this issue, email enquiry@pharmac.govt.nz
Please include as much information as you can about the product (presentation, brand, pharmacode) and who your wholesaler is.
Sign up to our email list for regular emails about supply issues and more(external link)


