Methenamine (hexamine) hippurate (Hiprex) tablets: Funding issue resolved
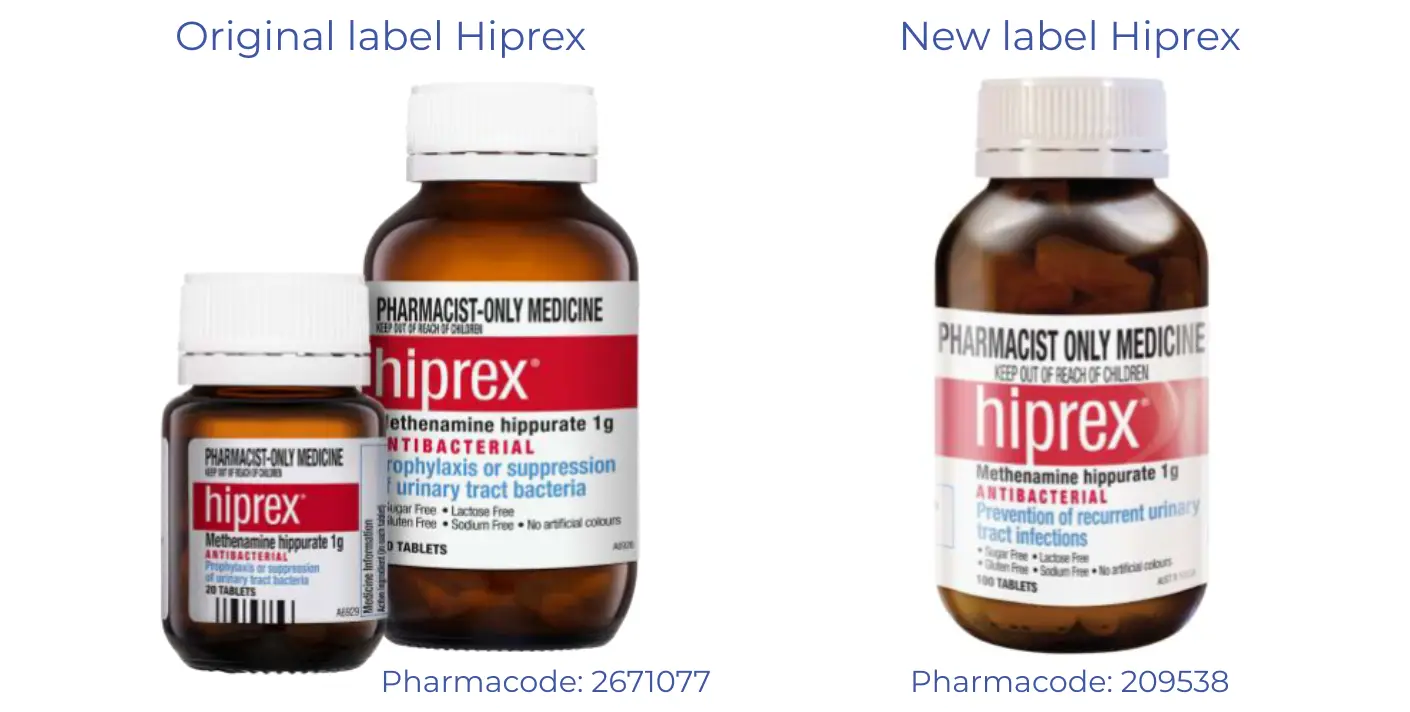
Due to a packaging change, there may be a funding issue with Hiprex tablets (Pharmacode: 2671077).
Some Hiprex tablets were not funded late 2025
We were alerted to a packaging change for Hiprex tablets, which included a change of Pharmacode.
The product with the new Pharmacode had been supplied by the Radiant Health since the end of November 2025. There may still be stock of the old Pharmacode in the supply chain.
The product with the new Pharmacode was not listed in the Schedule so pharmacies could not claim for it until it was listed.
Unfortunately, there was a misunderstanding with the supplier about when the change needed to be notified to Pharmac.
The previous Hiprex Pharmacode was 2671077, the new Pharmacode is 209538.
The new Hiprex has been reviewed by and registered with Medsafe.
It is funded again
The new Pharmacode was added to the Schedule from 1 January 2026. This was the earliest date we were able to action this.
What did this mean?
For any pharmacy dispensing Hiprex with the new Pharmacode before 1 January 2026, they needed to either charge the full price or the person will need a new prescription for a funded product.
We are sorry for any inconvenience caused. We have sought to put this right as soon as we learned of the problem. Unfortunately, we cannot list products on the Schedule outside of the regular monthly cycle.
Who to contact
If you have questions about this issue, email enquiry@pharmac.govt.nz
Please include as much information as you can about the product (presentation, brand, pharmacode) and who your wholesaler is.
Sign up to our email list for regular emails about supply issues and more(external link)
Pharmac is a government agency. We do not sell medicines or related products.


