Salbutamol with ipratropium (Duolin) nebuliser: Supply issue
Due to issues at the manufacturer, the supplier can no longer source Duolin nebulisers (Pharmacode: 2039281)
21 April 2026 | New alternative listed
A new alternative will be listed from 1 May 2026. The alternative is not Medsafe approved.
Affected product
The supplier can no longer supply this product:
- Chemical: Salbutamol 2.5 mg with ipratropium bromide 0.5 mg per vial
- Presentation: Nebuliser soln, 2.5 ml ampoule
- Brand: Duolin
- Pharmacode: 2039281
- Subsidy: $11.04
- Measure / Qty: per 20
Alternative listed 1 May 2026
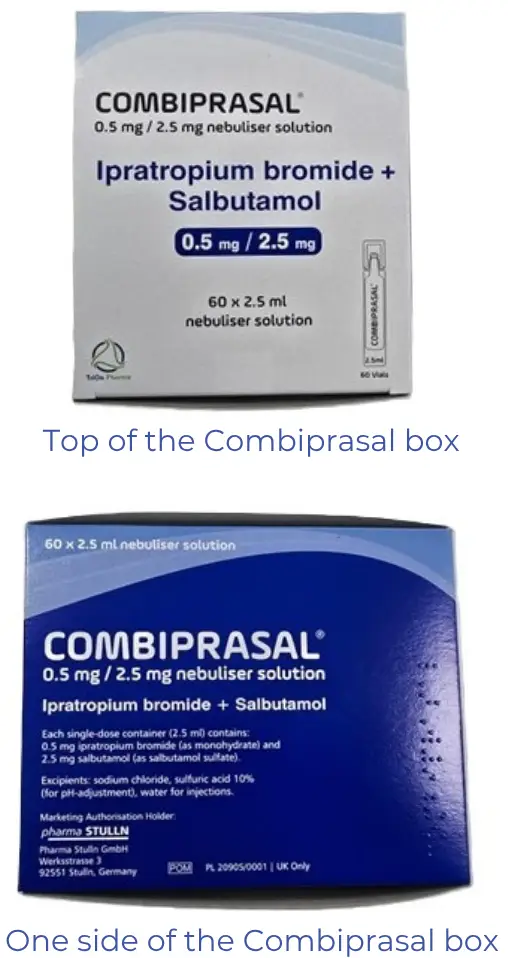
The supplier has sourced another alternative for Duolin nebules. It is not Medsafe approved and will need to be prescribed and dispensed in line with section 29A of the Medicines Act. Please note it comes in a 60-pack.
- Chemical: Salbutamol 2.5 mg with ipratropium bromide 0.5 mg per vial
- Presentation: Nebuliser soln, 2.5 ml vial
- Brand: Combiprasal
- Pharmacode: 2729512
- Subsidy: $96.50
- Measure / Qty: per 60
- Section 29 and wastage apply
Like the Cipla alternative, Combiprasal comes in vials.
Previous alternative listed 1 February 2026
The supplier, Rex Medical, secured stock of an alternative. This was listed in the Schedule from 1 February 2026. This medicine has not been approved by Medsafe. It will need to be prescribed and dispensed in line with section 29A of the Medicines Act. This product expires on 30 June 2026.
Differences
- Cipla S29 is a 30-pack, where Duolin is a 20-pack.
- Cipla S29 is a 3 ml vial where Duolin is a 2.5 ml ampoule.
- The vials are labelled "albuterol". This is the American spelling for salbutamol.
- Albuterol sulfate 3 mg is the same as salbutamol 2.5 mg
Listing details
- Chemical: Salbutamol 2.5 mg with ipratropium bromide 0.5 mg per vial
- Presentation: Nebuliser soln, 3 ml vial
- Brand: Cipla S29
- Pharmacode: 2719959
- Subsidy: $16.56
- Measure / Qty: per 30
- Section 29 and Wastage apply

- Duolin supply issue: Combiprasal brand change A5 flyer (April 26) [PDF 198 KB]
- Duolin supply issue Jan 2026 [PDF 169 KB]
Managing acute severe asthma
The Asthma and Respiratory Foundation has published algorithms for managing acute and severe asthma in adults and children.
Prescribing and supplying an unapproved medicine
Section 29A of the Medicines Act 1981 allows for medicines that are not Medsafe approved to be prescribed and supplied to people. However, unapproved medicines cannot be supplied under PSO.
We know supplying a medicine under section 29A is not ideal. In this case, however, this will allow people to continue to access an appropriate treatment.
We apologise for any inconvenience this causes.
Advice for prescribing under section 29 – BPAC website(external link)
What patients need to know about unapproved medicines – Healthify website(external link)
Who to contact
If you have questions about this issue, email enquiry@pharmac.govt.nz
Please include as much information as you can about the product (presentation, brand, pharmacode) and who your wholesaler is.
Sign up to our email list for regular emails about supply issues and more(external link)


