Factors for Consideration
The Factors for Consideration is the framework Pharmac uses when making funding decisions.
On this page
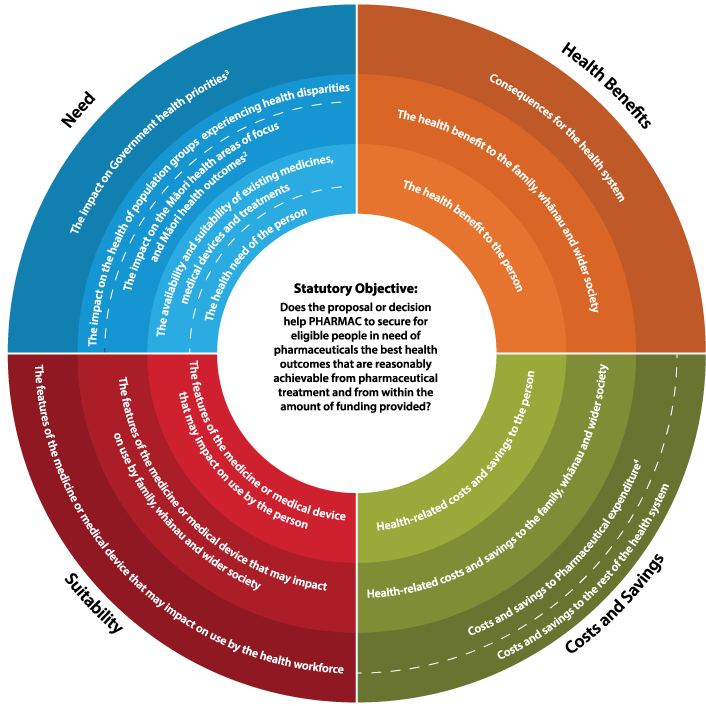
The four Factors for Consideration
- Need
- Health Benefits
- Costs and Savings
- Suitability
Each factor has 3 different aspects:
- the individual
- the family, whānau and society
- the health system.
Need
To work out what the level of 'need' is we consider the impact of the disease, condition or illness on the person, their family or whānau, wider society, and the broader New Zealand health system.
The health need of the person
- How unwell is a person compared to the average healthy New Zealander?
One major way in which we consider this is by comparing life expectancy and quality of life at full health and with the disease, condition or illness.
The availability and suitability of existing medicines and treatments
- What options are currently publicly funded to treat the population with this condition?
- How well do the current options work?
The 'suitability' of available medicines and treatments includes the practicality, effectiveness or appropriateness in the patient population group.
The impact on the health outcomes of population groups experiencing health disparities
What is the impact of the disease, condition, or illness on other population groups, including Pacific peoples, experiencing health disparities?
This Factor enables us to consider the impact of a decision on those that are facing health disparities as a result of an underlying disadvantage, separately from the illness itself. They may be characterised by ethnicity, culture, location, or socioeconomic status.
The impact on the Māori health areas of focus and Māori health outcomes
- What is the impact of the disease, condition or illness on Māori health outcomes?
Has the disease, condition or illness been identified as a Māori health area of focus in our Māori health strategy Te Whaioranga?
PHARMAC is committed to improving the health outcomes of Māori and being a great Te Tiriti/The Treaty partner. We work with Māori to identify specific health areas that are important to Māori communities.
Health System
- Is the disease, condition, or illness a Government health priority?
The Government chooses some health problems for the whole health sector to focus on. You can find the current health priorities in the Ministry of Health’s Statement of Intent, PHARMAC’s Statement of Intent, Output Agreement and/or Letter of Expectations.
Health Benefits
Health benefit is about the potential health gain from the medicine being considered.
The Health Benefit to the person
We consider, for example, if the treatment will make the person healthier, or help them live a longer life.
The Health Benefit to family, whānau and wider society
A medicine may have health benefits beyond the person receiving the treatment. For example, reducing antibiotic resistance will have positive health benefits for all New Zealanders.
Consequences for the health system
- If the medicine were funded, what would be the consequences for the health system?
PHARMAC’s decisions can have flow-on impacts for the rest of the health system. For example, if support services are required to administer a new treatment.
Costs and savings
We consider the costs and savings to the person and their family, whānau and to wider society. The cost and savings to the health system covers both the pharmaceutical budget and the wider health system.
Health-related costs and savings to the person
For example, the amount a person pays for a GP visit to be able to access the medicine.
Health-related costs and savings to the family, whānau and wider society
Funding a medicine may result in health-related costs and savings to family and whānau of the person receiving the treatment. For example, family and whānau may be caregivers, and a treatment may reduce the need for the level of care and the costs associated with this.
Costs and savings to pharmaceutical expenditure
- How would the funding of the medicine or related product impact on pharmaceutical expenditure?
- Would funding this medicine result in some savings due to people switching from another pharmaceutical that is already funded?
DHBs (District Health Boards) have limited funding available for pharmaceuticals used in the community or in hospital, so we need to consider the health outcomes that can be achieved from the limited amount of money available.
Costs and savings to the rest of the health system
Funding medicines or related products can have flow-on impacts for the rest of the health system (the health system refers to New Zealand’s health and disability system). For example, if a treatment can be given at home rather than in hospital it would free up a hospital bed for someone else to use.
Suitability
Suitability considers the non-clinical features of the medicine or medical device that might impact on health outcomes.
The features of the medicine or medical device that impact on use by the person
We may consider non-clinical features such as the size, shape and taste of a medicine, or its method of delivery (e.g. oral vs injection) that may affect health outcomes. For example, if a capsule is very large, some people may not be able to swallow it. This could affect their health outcomes.
The features of the medicine or medical device that impact on use by family, whānau and wider society
When family, whānau or members of wider society are the primary caregivers of a person receiving a medicine or medical device, the features of the medicine or medical device may affect their ability to administer the treatment. This in turn may affect the person’s health outcomes. For example, it may be easier for caregivers to give a sick person a pill than to give an injection.
The features of the medicine or medical device that impact on use by the health workforce
How the health workforce uses the medicine or medical device may affect the health of the person, for example, a medicine that is easy to use may reduce the likelihood of error or accident.
Putting the Factors to work
The factors help us assess each funding application against Pharmac's statutory objective. They help us answer the question:
Does the proposal or decision help Pharmac to secure for eligible people in need of pharmaceuticals the best health outcomes that are reasonably achievable from pharmaceuticals treatment and from within the amount of funding provided?
We will mainly use the Factors for Consideration when we’re making funding decisions, both decisions relating to treatments being listed on the Pharmaceutical Schedule, and also for decisions for individual patients through Pharmac’s Named Patient Pharmaceutical Assessment Policy.
When relevant, we also endeavour to use these Factors when making other decisions that relate to our statutory functions set out in the Pae Ora (Healthy Futures) Act 2022. This could include making decisions to support the responsible use of medicines.
Not every Factor may be relevant to every funding decision Pharmac makes
Some Factors may be more or less relevant (or may not be relevant at all) depending on the type and nature of the decision being made and, therefore, judgement is always required. Having the ability to exercise this judgement is critical to our role as it enables us to respond appropriately to a broad range of situations.
Every funding decision must be considered in relation to the amount of funding available
We must consider every funding decision in relation to how funding the given treatment may impact on the Combined Pharmaceutical Budget, District Health Board (DHB) hospital budgets and the overall Vote Health budget as applicable, for both the current financial year and over future years.
The Combined Pharmaceutical Budget is a fixed annual budget determined by the Minister of Health. Funding proposals for pharmaceuticals will be compared against one another using the Factors for Consideration, to determine which investments would result in the best health outcomes. Each funding proposal will then be ranked against all other funding proposals based on the results of this assessment. This is called the prioritisation process and the ranking helps us decide which investments to progress within the fixed annual budget.
Even though we do not currently manage budgets for hospital medicines and hospital medical devices, budget impact is still important for our role in these areas. It is important that we consider any financial implications of our decisions on DHBs. For hospital medicine funding decisions we have agreed with DHBs that new investments will not exceed the savings achieved on hospital medicines from Pharmac’s activities. For hospital medical devices, we've started contracting for products that DHB hospitals already buy, and are working to take on the full scope in a carefully planned way. Our work in hospital medical devices currently achieves direct savings for DHBs.
Pharmac seeks clinical advice on our funding decisions
For every funding decision we look at the strength and quality of the clinical evidence available. We also seek advice on the potential impact of funding the medicine in the New Zealand clinical treatment setting. Clinical evidence may be considered in detail by Pharmac’s expert advisory committee, the Pharmacology and Therapeutics Advisory Committee (PTAC), and where necessary the various specialist sub-committees of PTAC, and/or other external clinical specialists.


